 
In Nonpolar covalent compounds, the electrons are shared equally between the two atoms. In other words, these compounds do not have charged particles to transport electrons.īefore talking about the next property, let me tell you about polar and non-polar covalent bonds. Since there are no free moving ions or electrons, these compounds do not conduct electricity. 
What is the basic requirement to conduct electricity?Īnd if you recall, covalent compounds involve neutral molecules. Covalent compounds do not conduct electricity.Now, let me tell you about the third property. Note: Since the intermolecular forces are weak, covalent compounds are also soft and brittle (because they break or distort with force). This explains why they have a low melting and boiling point. So, a low amount of heat is required to break these bonds. Plus, the forces of attraction between these molecules are weak. Covalent compounds (generally) have low melting and boiling point.Īs I mentioned above, these compounds are made up of neutral molecules.Therefore, these compounds are usually volatile gases or liquids. This is the reason that forces of attraction between these molecules are weaker as compared to ionic compounds. Covalent bonding involves neutral molecules (having no electric charge).Here are the four properties that you should know: With this, it is time to move on and talk about the properties of covalent compounds. Ionic Bonding of sodium chloride (NaCl) | Best GuideĮxothermic and Endothermic reactions | Complete Guide This results in the formation of three covalent bonds, a so-called triple bond. In simple words, a nitrogen atom can fill its octet by sharing three electrons with another nitrogen atom. This means that a nitrogen atom needs three more electrons to become stable.Īs a result, two Nitrogen atoms share electrons with each other. If I talk about the Nitrogen atom ( N 7), it has seven electrons in total ( 5 in its valence shell). This example will help you understand this topic in a better way. To further understand this topic, let’s take a look at another example to understand this concept. This results in the formation of a water molecule (H2O). As a result, the oxygen atom shares electrons with 2 hydrogen atoms.A hydrogen atom has one electron and needs one more electron (according to the duplet rule).An oxygen atom has six electrons and needs two more electrons (according to the octet rule). 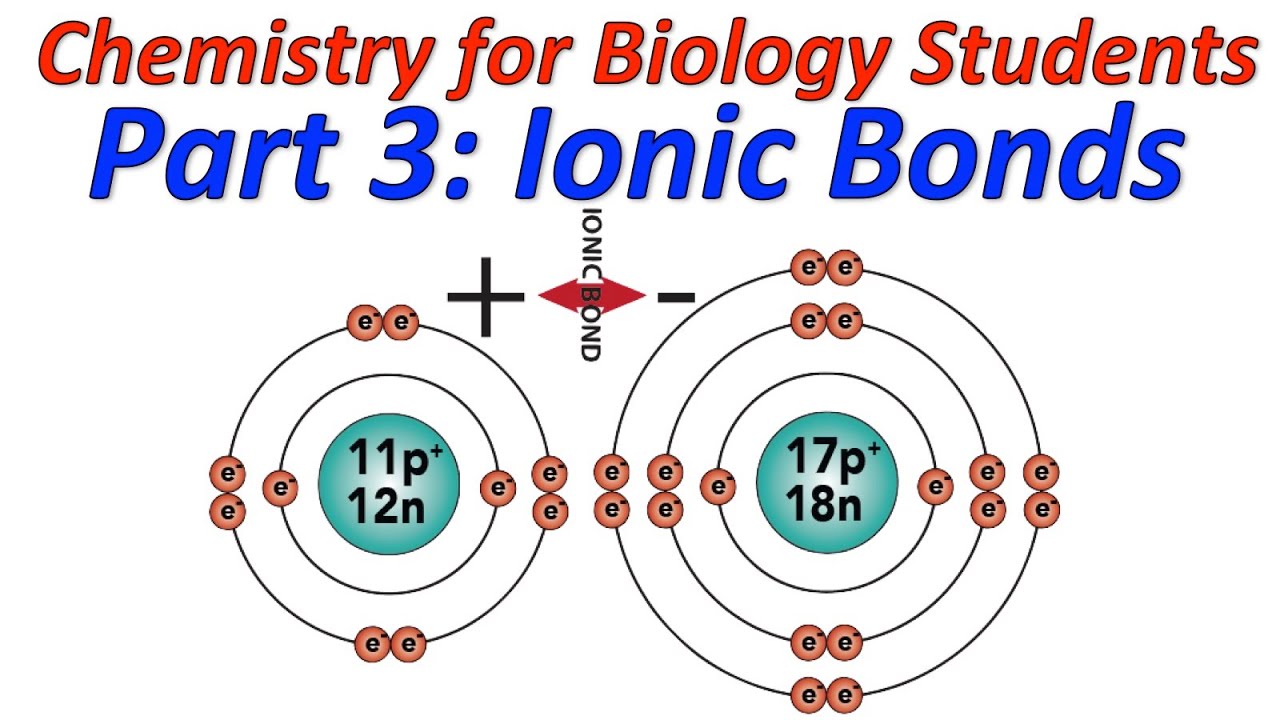
This is because of the unequal sharing of electrons (due to the difference in electronegativity) between oxygen and hydrogen. The Oxygen atom follows the octet rule in which the atom completes eight electrons in its valence (outermost) shell.Īs you can see in the image above, two hydrogen atoms and one oxygen atom share electrons with each other to become stable. Therefore, it needs two more electrons to become stable (achieve octet electronic configuration). Moving on, the Oxygen atom ( O 8) has six electrons in its outer shell. The Hydrogen atom follows the duplet rule that says: An atom is stable if it has two electrons in its outer shell. 
To become stable, the hydrogen atom needs one electron to achieve a duplet electronic configuration (the duplet rule). If I talk about hydrogen ( H 1), it has one electron in its outermost shell. If I talk about water (H2O), we will be talking about the first method.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
